Primary Angiosarcoma of the Breast: A Case Report
Article information
Abstract
Primary angiosarcoma of the breast is extremely rare and is observed in 0.0005% to 0.05% of primary breast tumor cases. The diagnosis of this tumor is difficult due to its undefined characteristics. Radiologic findings are often nonspecific and appear completely normal in one-third of patients with primary angiosarcomas. The prognosis is usually poor, and the treatment choices include mastectomy or wide excision. Radiotherapy and chemotherapy produce varying results. We report a patient with primary angiosarcoma of the breast to further our understanding of the characteristics of this tumor and facilitate the correct diagnosis of breast angiosarcoma.
INTRODUCTION
Angiosarcoma is a generic term that includes malignant sarcomas that originate from either the lymphatic or capillary endothelium, namely lymphangiosarcomas or hemangiosarcomas respectively. In the breast, these tumors are divided into two main categories depending on their origin, i.e., primary tumor in the breast is defined as primary angiosarcoma and secondary angiosarcoma originates after radiation therapy as cutaneous angiosarcomas [1]. Secondary angiosarcoma that arises in the irradiated breast after breast-conserving therapy is a well-established iatrogenic phenomenon that mainly involved the skin [1,2] and is being reported with increasing frequency. However, primary angiosarcoma of the breast that arises within the breast parenchyma is unusual.
Histologically, angiosarcomas are classified into three types: well-differentiated, moderately-differentiated, and poorly-differentiated. The degree of differentiation has a significant prognostic value, with regard to both local failures and metastases.
We report a case of a patient with well-differentiated angiosarcoma that arose primarily in the breast, with the aim of helping the medical community avoid the common trap of a benign differential diagnosis, achieve a better diagnosis of this cancer and improve our understanding of the proper management and prognosis of this cancer.
CASE REPORT
A 42-year-old woman, presented to our breast surgery clinic in March 2014 with complaints of a right breast lump, which had grown very slowly, and changed in color over the past year. She had no personal or family history of breast or ovarian cancer.
On physical examination, the lump was found to be at 12–1 o’clock position, and soft-to-firm in consistency, and discoloration and bruise-like staining were observed on the skin on the lump (Figure 1). On mammography, the lump presented diffuse and dense breast parenchyma, and the ultrasonography showed diffuse and poorly-delimited hyperechogenic infiltration in the upper inner portion of the right breast. This finding was very similar to edematous change (Figure 2). Due to the vague result of the imaging studies, the patient underwent contrast-enhanced breast magnetic resonance imaging (MRI). In the right upper inner breast, a lobulated, heterogeneously-enhanced, and irregular mass that measured approximately 6.8 cm in size was identified, and no evidence of axillary lymphadenopathy was observed. The mass displayed a delayed washout kinetic pattern (Figure 3). BexcoreⓇ (Medical Park Co., Yongin, Korea) assisted breast excision (vacuum assisted breast excision) was performed on the suspicious lesion in the right breast. The pathologic finding of the lesion was hamartoma and, follow-up was recommended.
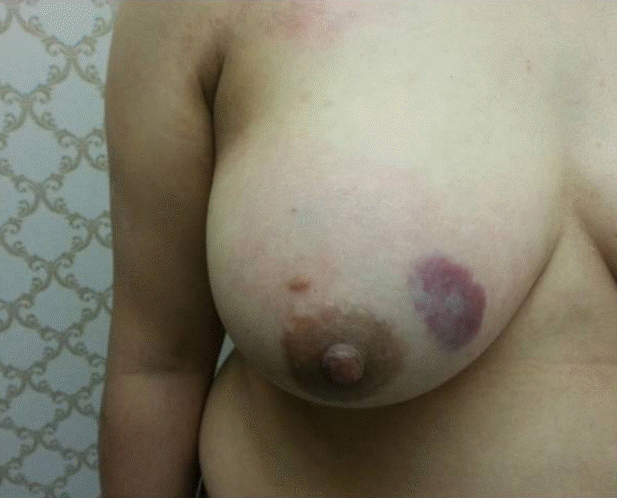
Physical findings. Soft-to-firm in consistencies, discoloration, and bruise-like staining is observed at the skin on the lump.

Mammography and ultrasonography findings. (A) On mammography, diffuse and dense breast parenchyma is observed. Otherwise, there are no specific findings. (B) Ultrasonography showed a diffuse and poorly-delimited hyperechogenic infiltration.
Magnetic resonance imaging findings. (A, B) A lobulated and heterogeneously-enhanced irregular mass is presented in the right upper inner breast. (C) The mass displays a delayed washout kinetic pattern.
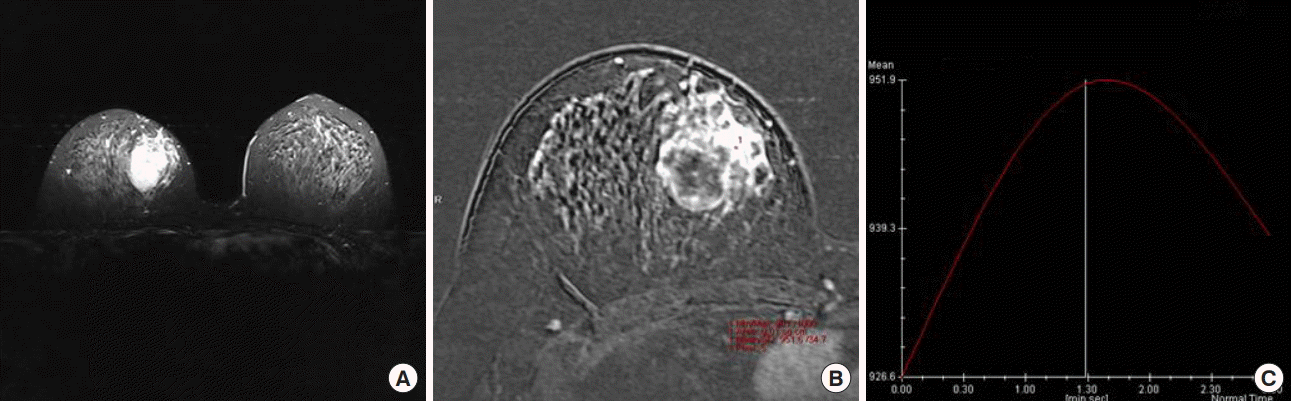
After 6 months, the patient revisited our clinic for discomfort of the right breast lump and complaints of breast pain. The ultrasonography showed a consistent edematous change and no significant new lesion. Therefore, follow-up recommended again, and on March 2015, the follow-up mammography revealed a solitary, ill-defined mass on the right breast (Figure 4). The ultrasonography showed a new cystic lesion without any other suspicious findings, excluding edematous change (Figure 5). Furthermore, the MRI revealed a larger, heterogeneously-enhanced T2 hyperintense mass lesion with delayed washout kinetic patterns in the upper inner portion of the right breast (Figure 6). We decided to perform a skin biopsy on the discolored portion, to obtain an accurate diagnosis, and the result was well-differentiated angiosarcoma. A modified radical mastectomy was performed, which we decided upon since the undefined margin and large mass size could interfere with partial mastectomy.
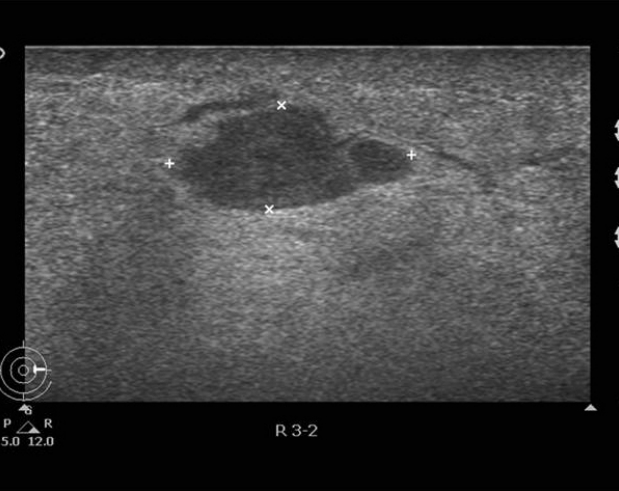
Follow-up ultrasonography. A cystic lesion is observed without other suspicious findings, excluding edematous change.

Follow-up magnetic resonance imaging (MRI) images. The MRI images show a large and heterogeneous (A, C), T2-hyperintense mass lesion (B) with a delayed washout kinetic pattern (D) in the upper inner portion of the right breast.
After mastectomy, at gross examination, the total size of the specimen was 18×9 cm. The tumor entirely replaced the upper quadrants, and measured 8.5×5.5×5 cm. The microscopic examination of the mass revealed many dilated vascular spaces that were lined and surrounded by spindle cell clusters, which are suggested of low grade angiosarcoma. There was involvement of the dermis in the skin, the surgical margins were tumor-free (Figure 7), and the stage was T4N0M0, consistently stage 3B. The positron emission tomography (PET) scan did not show any metastases; therefore, adjuvant chemotherapy (adriamycin combined with cyclophosphamide, four cycles) was prescribed to the patient. After that, she received radiation therapy, with total of 5,040 centigray (cGy) and 1,000 cGy on the chest wall and mastectomy site, respectively.
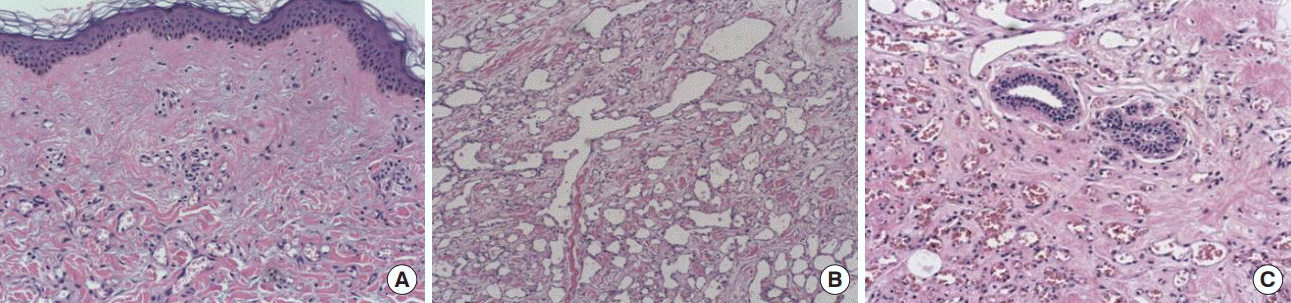
Pathologic results after mastectomy. The pathologic results after mastectomy show a small vascular proliferation in the dermis (A, H&E stain, ×40), marked- and well-differentiated vascular proliferation (B, H&E stain, ×40), and significant periductal vascular proliferation (C, H&E stain, ×100).
On January 2018, the follow-up studies (including MRI and PET) showed no complications or recurrence.
DISCUSSION
Mammary sarcomas are a heterogeneous group of malignant neoplasms that arise from the mammary stroma. Angiosarcomas are the most common sarcoma of the breast, but are still relatively rare. The neoplasm is of vascular origin, and the frequency of this rare tumor is <1% of all sarcomas, and the incidence is 0.0005% to 0.05% of all malignant neoplasms of the breast [2], and approximately 8% of all primary breast tumors. The first case was reported by Schmid in 1887.
While primary angiosarcoma arises without a recognized associated factor, it is not clear whether secondary angiosarcoma are induced by edema or radiation or have a multifactorial origin. Secondary angiosarcoma are usually induced by specific characteristics: location in previous radiation fields and latency after radiation therapy. There are no distinct clinical or histologic differences between primary and secondary angiosarcomas [1]; however, Fraga-Guedes et al. [2] reported the differences between primary and secondary breast angiosarcoma. These investigators suggested that secondary angiosarcomas are more likely to have high-grade pathologies than primary angiosarcomas and poorer prognoses compared to primary angiosarcomas. While this article is interesting, it is limited by the small number, of patients (only 28). Many studies have described that primary angiosarcomas, but not secondary angiosarcomas, usually occur in younger women (20–50 years) [1,2].
Angiosarcomas may have an insidious clinical onset, presenting as a painless, often discrete palpable mass that grows rapidly [1]. However, our patient had a palpable mass that grew very slowly for two years; therefore, we think that rapid growth is not typical. Fraga-Guedes et al. [2] found that patients with primary angiocarcinoma were more likely to present with the complaint of a mass, while patients with secondary angiocarcinoma were more likely to present with a complaint of a rash. Our patient also reported discomfort from the mass, rather than from skin rash. Some patients complain of a painful mass with tenderness, and approximately 2% of patients may present with initial diffuse enlargement of the breast [1]. A bluish-red discoloration of the overlying skin due to tumor infiltration is a frequent symptom; however, involvement of the underlying muscles is rare [1]. Nipple retraction, discharge, or axillary node enlargement are generally absent.
Radiographically, breast angiosarcomas exhibit no pathogenic features. They often appear as ill-defined masses on mammograms or without significant findings. Calcifications can be seen, but differ from those seen in case of breast carcinomas [3]. Diagnosis prior to surgery, either by fine-needle aspiration (FNA) or core needle biopsy, is always difficult. In our case, we also could not obtain a definitive diagnosis through imaging studies and FNA before skin biopsy. Ohta et al. [4] cited Chen’s study that reported 39% (22 of 57 cases) of patients reported before 1967 and 33% (10 of 30 cases) of those reported after 1967 had been misdiagnosed as having benign lesions at initial biopsy. Additionally, Rainwater et al. [5] reported that 50% of patients (10 of 20 cases) were initially misdiagnosed as having benign lesions.
Mammary angiosarcomas should be differentially diagnosed from benign hemangiomas, phyllodes sarcomas, stromal sarcomas, metaplastic carcinomas, fibrosarcomas, liposarcomas, squamous cell carcinomas with sarcomatoid features, myoepitheliomas, fibromatoses, and reactive spindle cell proliferative lesions [4]. CD31 is a sensitive marker for this class of cancers, and CD34 positively ranges from 40% to 100%. Immunostaining for factor VIII has been consistently positive, with a stronger staining of malignant cells than has been observed in traditional nonepithelioid vascular sarcomas [6]. These markers could help the accurate diagnosis of angiosarcoma.
Pathologically, these tumors are divided into three groups according to the classification proposed by Donnel et al. [7]. Well differentiated (grade I) tumors consist of anastomosing vascular channels that invade the surrounding breast tissue. Moderate differentiated (grade II) tumors have more solid neoplastic vascular growth and an increased mitotic rate. Poorly differentiated (grade III) lesions have obvious sarcomatous areas, and areas of necrosis, hemorrhage, and infarction.
The prognosis is generally poor and based primarily on tumor size, tumor grade, completeness of surgery, and performance status. However, the factors related to the prognosis of primary breast angiosarcoma have remained controversial. One study reported that patients with higher-grade lesions at pathologic evaluation were significantly more likely to develop recurrent disease and have unfavorable prognosis [8]. In the study of Rosen et al. [9], the 5-year disease-free survival rate for low grade tumors was high as 76% and up to 70% for intermediate grade tumors. Additionally, the 5-year survival rate for high-grade tumors is approximately 15%. Otherwise, Rainwater et al. [5]. reported that in the 20 patients who were studied, the histological grading did not seem to correlate with mean patient survival. Furthermore, the gross tumor size did not correlate with survival. However, other studies have reported that overall survival and recurrence-free survival significantly associated only with primary tumor size.
Primary angiosarcoma is an aggressive neoplasm with a tendency for local recurrence and distant metastasis. The commonest sites of metastasis are the bone, lung, liver, brain, ovary and skin. Lymph node involvement is present in only 6% to 8% of patients. Angiosarcoma have a high mortality rate, and only 10% to 21% of patients remain disease-free after 5 years [10].
There is no established standard treatment for angiosarcomas. Surgical resection is recommended as the primary treatment, and mastectomy is the mainstay of treatment. However, if complete local excision with histologically clear margins can be performed, conservative surgery is possible. One study reported that conservative surgery versus mastectomy did not lead to statistically significantly different outcomes [10]. Hematogenous dissemination is the rule, making axillary lymph node dissection unnecessary, except in case of definite lymph node metastasis.
Although patients with well-differentiated tumors do not appear to derive benefit from adjuvant chemotherapy, the effects for patients with disseminated disease have been reported; however, the results have been questionable. In advanced breast angiosarcoma and most other subtypes of soft tissue sarcomas, the classical treatment and standard-first line therapy is based on a doxorubicin-containing regimen. Some retrospective studies have identified the efficacy of weekly paclitaxel as a treatment for metastatic or locally-advanced angiosarcoma [11]. However, there is still no general agreement about a standardized therapy for patients who progress while on anthracycline.
Postoperative irradiation can be used to improve local control, especially in patients with tumors larger than 5 cm and with positive surgical margins. Microscopic tumoricidal dose (50 Gy) to the whole beast, and at least 60 Gy to the tumor bed, is usually used [12].
Immunotherapy may also play a role in treatment of breast angiosarcomas. Burrows et al. [13] suggested that the antibody, which is directed against the endoglin molecule, is a promising agent for the vascular targeting approach, since endoglin is overexpressed on tumor-associated endothelial cells, and not on normal endothelial cells.
Some have investigated an approach that involves attacking the proliferative endothelium. Close interactions between endothelial cells and the blood stream appears to make the vasculature a practical target for tumor therapy. New agents against angiogenesis, such as bevacizumab or rapamycin, might also be useful against this tumor [14].
In conclusion, primary mammary angiosarcoma is a rare neoplasm of the breast that affects a younger female population, compared to breast carcinomas, and has aggressive clinical behavior. Difficult differential diagnoses, due to atypical characteristics, can delay management; the accurate diagnosis of angiosarcoma, and, in particular, the differentiation from primary breast carcinoma, are important for optimal therapy.
Notes
The authors declare that they have no competing interests.
