Phosphorylated S6 Kinase-1 as Predictive Marker of Lapatinib Efficacy in Human Epidermal Growth Factor Receptor 2-Positive Metastatic Breast Cancer Patients
Article information
Abstract
Purpose
The 40S ribosomal protein S6 kinase-1 (S6K1) is a crucial downstream effector of the PI3K/AKT/mTOR pathway. S6K1 overexpression is found in 10% to 30% of breast cancers and is associated with aggressive disease and poor prognosis. Herein, we investigated the relationship between the expression of phosphorylated S6K1 (p-S6K1) and efficacy of lapatinib in patients with human epidermal growth factor receptor 2 (HER2)-positive metastatic breast cancer.
Methods
We retrospectively analyzed the data of 36 patients with HER2-positive metastatic breast cancer treated with lapatinib between January 2010 and September 2014. The p-S6K1 expression status of the primary tumor was assessed via immunohistochemistry using a mouse monoclonal antibody.
Results
Fourteen of the 36 patients (38.9%) had p-S6K1-positive tumors. The median progression-free survival (PFS) of patients with p-S6K1-positive tumors was significantly longer than that of patients with p-S6K1-negative tumors (13.4 months vs. 7.1 months, p=0.025). In multivariate analysis, p-S6K1 positivity remained an independent, favorable predictive factor for PFS (hazard ratio, 0.32; 95% confidence interval, 0.11–0.97; p=0.044).
Conclusion
The high expression of p-S6K1 was significantly associated with prolonged PFS, suggesting that p-S6K1 can be a potential biomarker for predicting the efficacy of lapatinib in patients with HER2-positive metastatic breast cancer.
INTRODUCTION
Overexpression of the human epidermal growth factor receptor 2 (HER2), which is attributable to HER2 (erbB2/neu) gene amplification, is identified in approximately 20% to 25% of all cases of breast cancers [1]. HER2 activation is achieved through either homodimerization with other HER2 in a ligand-independent fashion or heterodimerization with other members of the HER family as a result of ligand stimulation. HER2 activation leads to subsequent activation of the PI3K/AKT and Ras/ERK/MAPK signaling pathways [2]. Activation of these pathways promote cell proliferation, evasion of apoptosis, angiogenesis, and invasion, leading to tumorigenesis and progression [3]. Consequently, HER2 overexpression in breast cancer correlates with a more aggressive disease course [4]. Therefore, numerous efforts have been made to develop HER2-targeted therapeutics, and some of the successful efforts has revolutionized the treatment of HER2-positive breast cancer and led to significant improvement in clinical outcome. Lapatinib, one successful example, is an oral dual tyrosine kinase inhibitor that competes with ATP binding sites of epidermal growth factor receptor and HER2, resulting in inhibition of proliferation and activation of downstream signal transduction that includes MAPK and PI3K/AKT/mTOR pathways [5]. It was approved in 2007 to be used in combination with capecitabine (Xeloda®) for the treatment of advanced or HER2-overexpressing metastatic breast cancer that has progressed following prior therapy with trastuzumab, anthracycline, or taxane [6]. Despite the reported and proven benefits of lapatinib in patients with HER2-overexpressing metastatic breast cancer, only 20% to 35% of these patients respond to lapatinib, and the vast majority of patients failed to respond to treatment with lapatinib owing to de novo resistance. Even responders eventually develop acquired resistance within a year [7,8]. Therefore, a clear understanding of the mechanisms of lapatinib resistance will help us determine which patients would benefit the most from lapatinib treatment. Importantly, factors causing lapatinib resistance may serve as molecular therapeutic targets for overcoming lapatinib resistance.
Dysregulation of the PI3K/AKT/mTOR signaling pathway through the loss of the PTEN phosphatase tumor suppressor function or somatic activating mutations in PI3K3CA have consistently been implicated in the de novo resistance to trastuzumab, which is an HER2-targeting monoclonal antibody [3,9-14]. However, studies on the correlation between lapatinib sensitivity and activation of PI3K/AKT/mTOR signaling pathway reported conflicting results, showing inverse or no relation between the two [13-16].
The 40S ribosomal protein S6 kinase-1 (S6K1) is a crucial downstream effector of PI3K/AKT/mTOR signaling, and its phosphorylated form is used as a surrogate marker for PI3K/AKT/mTOR activity in laboratory and clinical research studies [17-25]. In this study, we sought to explore the relationship between the expression of phosphorylated S6K1 (p-S6K1) and response to lapatinib treatment in patients with HER2-positive metastatic breast cancer.
METHODS
Study patients and database
In this study, we retrospectively reviewed the medical records of 36 patients treated with lapatinib-based therapy between January 2010 and September 2014. The principal eligibility criteria for this study included patients with confirmed HER2 positivity, history of lapatinib-based therapy for metastatic breast cancer, and availability of p-S6K1 expression status via immunohistochemical staining. All patients received lapatinib (1,250 mg/day continuously) plus capecitabine (1,000 mg/m2/day, 2-week-on and 1-week-off) until disease progression, death, or intolerable toxicity. Clinical information was retrieved retrospectively from the database at the Korea Cancer Center Hospital. The Korea Cancer Center Hospital database is a prospectively maintained, web-based system that includes information on all patients with breast cancer that have undergone breast cancer surgery at Korea Cancer Center Hospital since 1983. To date, more than 5,000 patients with breast cancer have been registered in this database [22]. This study complies with the current laws of South Korea, including ethics approval. This study was approved by the Korea Cancer Center Hospital Institutional Review Board (K-1502-002-027). Written informed consent has been waived.
Immunohistochemistry and p-S6K1 scoring
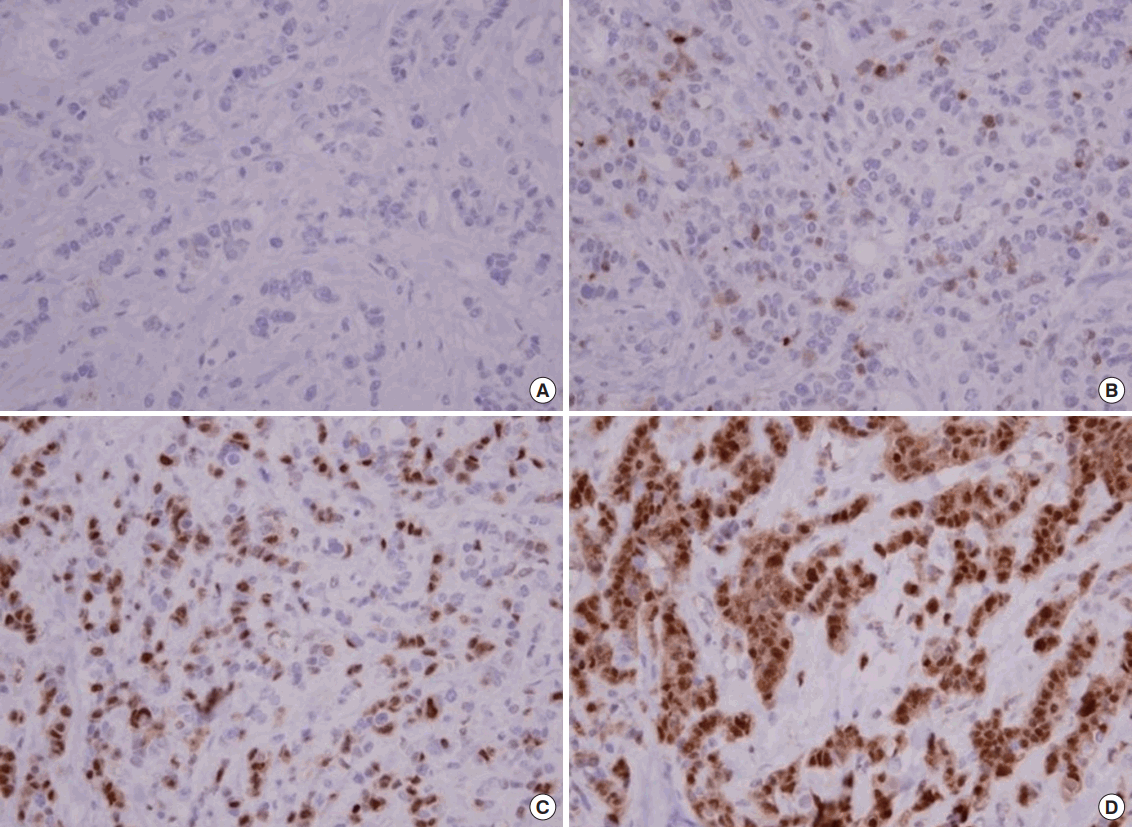
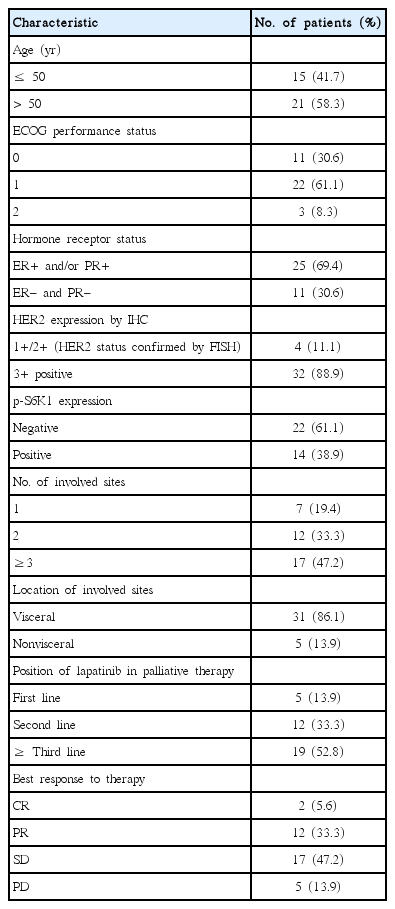
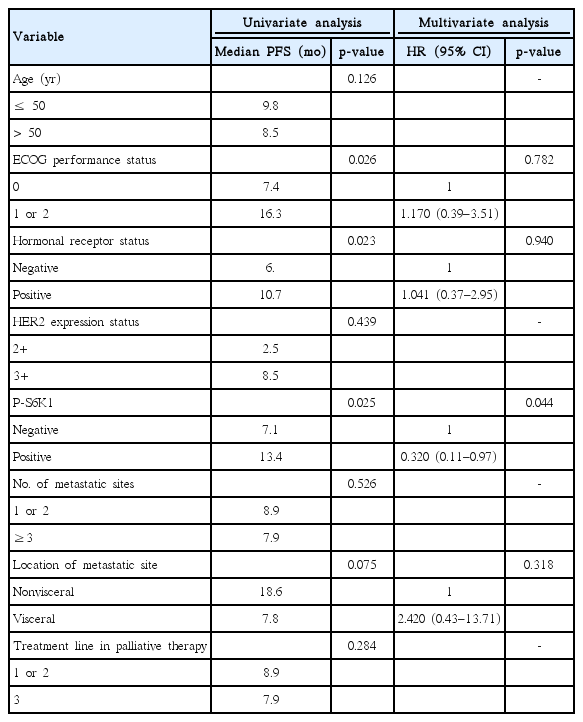
The p-S6K1 expression status of the primary tumors was detected via immunohistochemistry using the mouse monoclonal antibody to p-S6K1 (Cell Signaling Technology Inc., Danvers, USA; dilution 1:50). The results of the staining were evaluated using a combined intensity and percentage positive scoring method and were scored as follows: 0 (no staining or staining in <10% of the tumor cells), 1+ (weak to moderate staining in 11%–20% of the tumor cells), 2+ (moderate to strong staining in 21%–50% of the tumor cells), and 3+ (strong staining in >50% of the tumor cells). A score of 2+ or 3+ was regarded as positive, while the other scores were regarded as negative (Figure 1).
Statistical analysis
The chi-square test and Fisher exact test were used to determine the correlation between p-S6K1 status and other clinicopathological characteristics. Response to treatment with lapatinib was assessed using the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines criteria version 1.1. Progression-free survival (PFS) was defined as the time from the initiation of lapatinib-based treatment until the first documented disease progression. Survival analyses were performed using the Kaplan-Meier method. A univariate analysis was performed, and the log-rank test was used to evaluate the effects of several clinicopathological factors on PFS. Multivariate analysis was performed through a Cox proportional hazards regression model. In all tests, a p-value <0.050 was considered statistically significant.
RESULTS
Clinicopathologic characteristics and p-S6K1 expression
A total of 36 patients with HER2-positive metastatic breast cancer were included in this analysis. All patients were pretreated with anthracyclines, taxanes, and trastuzumab. The clinicopathological characteristics are presented in Table 1. The median age at the time of treatment with lapatinib was 52 years (range, 32–60 years). A total of 25 of the patients (69.4%) had estrogen receptor (ER)- and/or progesterone receptor-positive tumor, and 29 (80.5%) had more than two metastatic diseases, most of which are visceral metastasis. The most frequent second-line treatment used for metastatic breast cancer was lapatinib plus capecitabine (31, 86.1%). According to the criteria described above, positive p-S6K1 expression was found in 14 patients (38.9%); of them, 11 (30.6%) and three (8.3%) had 2+ and 3+ tumors, respectively (Figure 1). The expression status of p-S6K1 was not associated with any conventional clinicopathological parameters, such as Eastern Cooperative Oncology Group (ECOG) performance status, age, hormone receptor status, HER2 protein expression status, or the number of metastatic sites (all p>0.050).
Correlation of p-S6K1 expression with progression-free survival
During the follow-up period, 31 patients experienced disease progression (91.8%), and 10 patients died from their disease (29.4%). The median follow-up period was 14.9 months (range, 2.1–46.7 months). The median PFS of all 36 patients was 8.9 months (95% CI, 6.5–11.1 months). We found that the median PFS of patients with p-S6K1-positive tumor was significantly longer than that of patients with p-S6K1 negative tumor (13.4 months vs. 7.1 months, p=0.025 by log-rank test) (Figure 2). Table 2 presents a univariate analysis of clinicopathological variables that might influence PFS including ECOG performance status, age, hormone receptor status, HER2 expression, metastatic sites, number of metastatic sites, and use of lapatinib in palliative therapy. ECOG performance status, hormonal receptor, and p-S6K1 expression were statistically associated with PFS. When these three variables plus the type of metastases (i.e., visceral, nonvisceral; p=0.075) were entered into Cox proportional hazard model to evaluate the independent value of p-S6K1 status and other variables predicting PFS, only p-S6K1 expression was found to be an independent factor for PFS in patients with HER2-positive metastatic breast cancer treated with lapatinib.
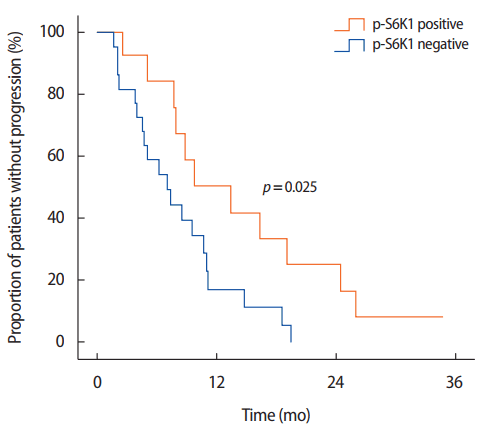
Cumulative Kaplan-Meier survival curve showing that patients with phosphorylated ribosomal protein S6 kinase-1 (p-S6K1)-positive tumor had significantly longer progression-free survival than patients with p-S6K1 negative tumor.
Correlation of p-S6K1 expression with response to therapy
A total of two (5.6%) and 12 (33.3%) patients achieved complete and partial response, respectively. Stable disease as the best response was achieved in 17 patients (47.2%), and disease progression despite treatment with lapatinib was seen in five patients (13.9%).
DISCUSSION
In this retrospective study, we evaluated the relationship between p-S6K1 expression status and PFS in patients with HER2-positive metastatic breast cancer who received lapatinib-based therapy. Our results demonstrated that patients with elevated p-S6K1 level had significantly longer PFS than those with relatively low p-S6K1, indicating that tumors expressing p-S6K1 have better treatment response to lapatinib.
S6K1 is a mitogen-activated serine/threonine kinase and a key downstream effector of the PI3K/AKT/mTOR pathway, which is a crucial intracellular signaling pathway in numerous cellular activities, including cell proliferation, differentiation, and survival. S6K1 overexpression was found in 10% to 30% of primary breast cancers and is associated with aggressive disease and poor prognosis. In addition, dysregulation of the PI3K/AKT/mTOR pathway is implicated in cancer progression, metastasis, and resistance to anticancer treatments such as chemotherapeutic agents, endocrine therapies, and radiotherapy [17-23,25]. PIK3CA mutation and PTEN loss, a negative regulator of PI3K signaling, are the most common genetic alterations in this pathway. Both genetic alterations can lead to downstream activation of the PI3K/AKT/mTOR pathway, and thus contribute to poor clinical outcome [3,22-26]. The relationship between loss of PTEN or PIK3CA mutation and HER2-targeted therapy in HER2-positive breast cancer has been demonstrated in both preclinical and clinical studies. As for trastuzumab, PTEN loss and PIK3CA mutation are consistently reported to be associated with poor treatment response to trastuzumab due to the inability of this drug to block PI3K signaling [3,9-14]. Meanwhile, results regarding the correlation between activation of the PI3K/AKT/mTOR pathway and lapatinib response are conflicting. Some preclinical studies demonstrated that the loss of PTEN and/or PIK3CA mutation are implicated in resistance to lapatinib in HER2-positive breast cancer cell lines and xenograft models. Some other clinical studies also reported that PI3K activation resulting from loss of PTEN or PI3K mutation was associated with better response in patients treated with lapatinib-plus-capecitabine [13,16]. By contrast, a phase II study of neoadjuvant HER2-targeted therapy demonstrated that patients with low PTEN tumor exhibited a significantly high pathologic complete response rate [14]. Because p-S6K1 has been reported to be significantly associated with PTEN loss or PIK3CA mutation, p-S6K1 is regarded as a surrogate marker of PI3K/AKT/mTOR pathway activity [10,24,25]. As such, we evaluated the relationship between p-S6K1, which represents the activation of this pathway, and response to lapatinib treatment in patients with HER2-positive metastatic breast cancer to determine whether PI3K/AKT/mTOR pathway activation is associated with treatment response to lapatinib. Interestingly, our results showed that the high level of p-S6K1 is significantly associated with better response to lapatinib in contrast to general knowledge that p-S6K1 expression is correlated with poor clinical outcome as seen in resistance to chemotherapy or endocrine therapy. Some studies investigated the relationship between S6K1 and lapatinib response. One is a small pilot study that assessed the biologic effects of lapatinib on tumor growth and survival pathways and reported that the elevated levels of S6K1 was associated with better clinical response [27]. Another is an in vitro study that examined the basal level of S6K1 based on IC50, a drug concentration causing inhibition of cell proliferation by 50%, after dividing HER2-amplified cell lines into the two groups of lapatinib-sensitive and lapatinib-resistant cell lines. The result of this study showed that basal S6K1 expression was higher in lapatinib-sensitive cell lines [28]. Vazquez-Martin et al. [29] treated MCF-7/HER2, a HER2-positive cell line, with trastuzumab and lapatinib and measured the level of S6K1. They reported that lapatinib treatment significantly decreased the phosphorylation of S6K1, whereas trastuzumab failed to deactivate S6K1, suggesting that S6K1 is a potential biomarker predicting response to lapatinib. Collectively, these studies, including our study, imply that high levels of S6K1 may be associated with better lapatinib treatment response.
S6K1 directly phosphorylates ER-α on Ser167, which increases ER-α transcriptional activity and ER-dependent breast cancer cell proliferation. Although p-S6K1 expression may be correlated with poor prognosis in patients with ER-positive tumors [30], we could not conduct a subgroup analysis according to the ER status due to the small sample size.
To our knowledge, no clinical studies addressing the association between p-S6K1 and lapatinib response have been published. Although conflicting results from other studies suggest more complicated mechanisms on lapatinib resistance, our results show that an elevated p-S6K1 level is associated with long PFS following lapatinib treatment, implying that p-S6K1 status may be an independent predictor for responsiveness for lapatinib treatment.
However, our study has several limitations. The main limitation of this study is that this is a retrospective study with a relatively small sample size, and further research that examines a large group of patients is warranted. A second limitation is that we cannot exclude the possibility of change of p-S6K1 level between the period of initial biopsy and that of lapatinib treatment, considering the effects of heavy pretreatment received by most patients. Third, since patients were treated with lapatinib-plus-capecitabine regimens, we must consider the possibility of capecitabine-mediated effect. Lastly, the cutoff in positive cells defining a tumor with p-S6K1 overexpression often varies according to the investigators, which may lead to biased conclusions.
Despite these limitations, to our knowledge, this is the first study to specifically investigate the clinical correlations between p-S6K1 status and treatment response to lapatinib, and we found a significantly positive association between the two. Moreover, as p-S6K1 is readily detectable via immunohistochemical staining in biopsy samples and highly specific antibodies to p-S6K1 are already commercially available, p-S6K1 can be proposed as a novel biomarker for predicting response to lapatinib treatment in patients with HER2-positive metastatic breast cancer.
In conclusion, we identified p-S6K1 as a potential valuable marker to predict successful outcome of lapatinib treatment. This is the in vivo study that identified p-S6KI as potential biomarker for predicting the treatment efficacy of lapatinib in patients with HER2-positive metastatic breast cancer.
Notes
The authors declare that they have no competing interests.
Acknowledgements
This study was supported by a grant from the Korea Cancer Center Hospital (grant number: 1711045543; 1711045540/50472-2017).